Effect of Nitrogen on the Corrosion Resistance of 6Mo Super Austenitic Stainless Steel
Abstract
1. Introduction
The 6Mo super austenitic stainless steel (SASS) is rich in a large amount of
chromium, molybdenum and nickel. These elements facilitate the spontaneous
formation of a continuous dense passive film on the surface, which significantly
enhances the corrosion resistance and mechanical properties of stainless steel
[1,2,3]. Currently, 6Mo SASS is widely used in aggressive environments such as
the petrochemical industry, flue gas desulfurization, seawater desalination
systems, and pulp mill bleaching systems. The research on the performance of
6Mo SASS has also attracted more and more researchers’ interest. For instance,
Sandra et al. [4] found that 6Mo high-nitrogen grade UNS S31266 showed
excellent corrosion resistance in acidic media containing chloride. Behrens et al.
[5] found that the combination of high nickel content and nitrogen in 6Mo super
austenitic stainless steel resulted in favorable low sigma solvent temperatures, and
high chromium and molybdenum content resulted in resistance to common forms
of corrosion and localized corrosion in chlorine-containing media. Ren et al. [6]
studied the effect of boron addition on the oxide skin of S31254 super austenitic
stainless steel in high-temperature air, and the results showed that the addition of
B could inhibit the diffusion of Mo to the surface and promote the diffusion of Cr to
the surface to form a dense Cr2O3 film. Yang et al. [7] found that the grain
boundary segregation of B and Ce had a significant inhibitory effect on precipitates,
and the addition of B and Ce could greatly improve the corrosion resistance of
S31254 SASS. Dou et al. [8] investigated the passivation performance of S31254
SASS in simulated condensate after flue gas desulfurization at different
temperatures. The results revealed that acidic condensation accelerated the
preferential dissolution and subsequent precipitation of Fe and the oxidation of Mo,
resulting in the formation of a mixed Cr-Fe-Mo oxide/hydroxide surface layer. Chen
et al. [9] studied the effect of Cl− on the corrosion and passivation behavior of the
same steel in blast furnace gas (BFG) electrolyte. The results indicated that the
passivation ability of S31254 SASS was greater under low-chloride ion
concentrations compared to high concentrations. In addition, Liu et al. [10] reported
that the pH value significantly influenced the passivation behavior of S31254 SASS
in NaCl solution. In weakly acidic and strongly acidic solutions, the main
components of the passive film outer layer were iron oxide and Cr(OH)3,
respectively. Li et al. [11] demonstrated that the corrosion resistance of 6Mo SASS
initially improved but later weakened with increasing copper content.
The influence of nitrogen on the properties of stainless steel has drawn significant
attention from researchers. Nitrogen not only enhances the re-passivation ability
of stainless steel but also plays a crucial role in improving the passive film [12,13].
For example, Aamani et al. [14] observed that nitrogen addition leads to grain
refinement and an increased proportion of low-energy boundaries, thereby
enhancing corrosion resistance. Loable et al. [15] reported a synergistic effect
between nitrogen and molybdenum on the pitting potential. Moreover, Dai et al.
[16] demonstrated that nitrogen significantly enhances the corrosion resistance of
316 L stainless steel in a thiosulfate-chlorine solution by promoting the formation
of chromium oxide and iron oxide in the passive film. Gao et al. [17] reported a
higher proportion of stable oxides, such as chromium oxide, in the passivation film
of high-nitrogen stainless steel. The results also revealed that the content of
ammonium ions is higher in high-nitrogen stainless steel, effectively inhibiting
passive film breakdown and pitting. Furthermore, Zhang et al. [18] investigated the
impact of nitrogen on precipitation behavior, intergranular corrosion resistance,
and mechanical properties of S32654 SASS. The results indicated that S32654
with medium nitrogen content exhibits the smallest total amount of precipitates
after aging treatment and the best intergranular corrosion resistance.
Nevertheless, the influence of nitrogen content on the corrosion resistance and
passive film structure of 6Mo SASS remains unclear.
2. Experimental Procedure
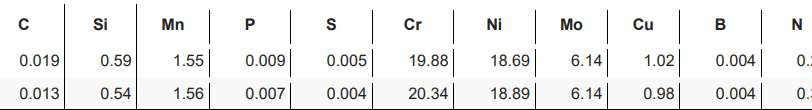
Material Preparation
Microstructure Characterization
Electrochemical Experiment
XPS Analysis
3. Results and Discussion
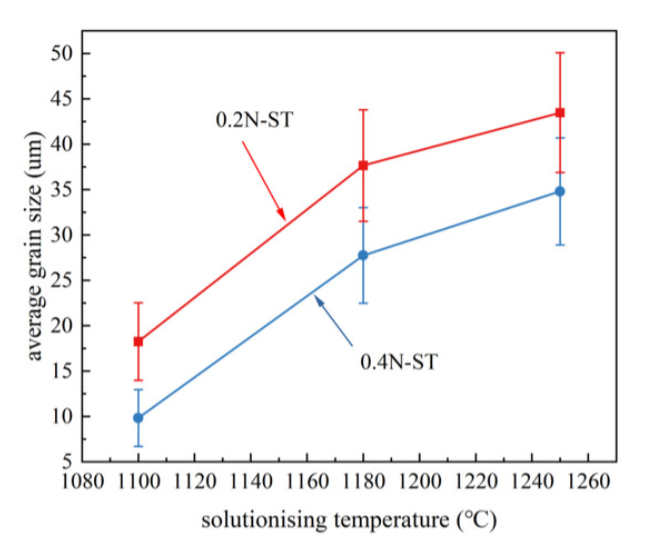
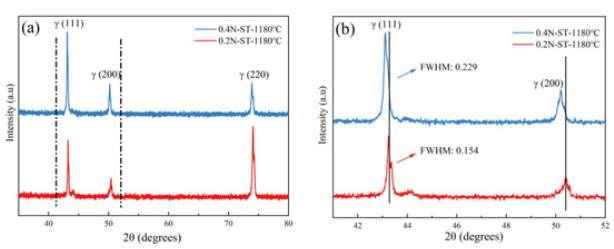
Microstructure Characterization


3.2. Polarization Curve Measurements
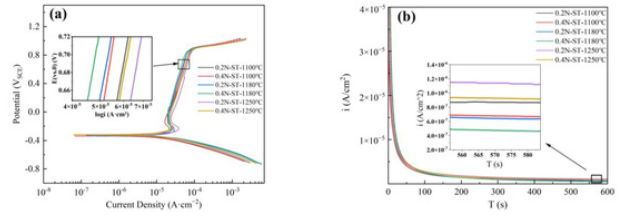
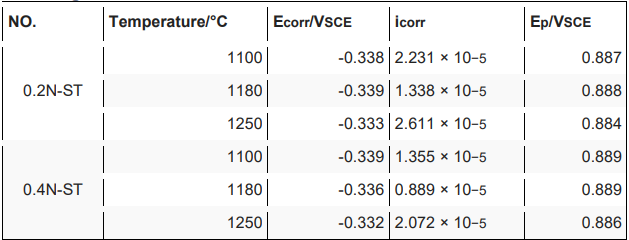
Electrochemical Impedance Spectroscopy Measurements
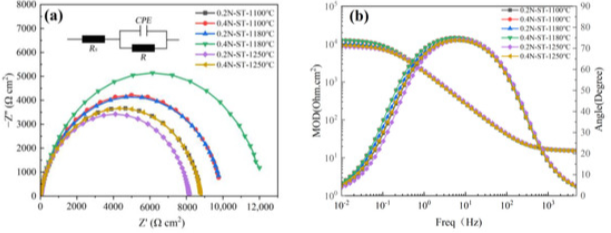
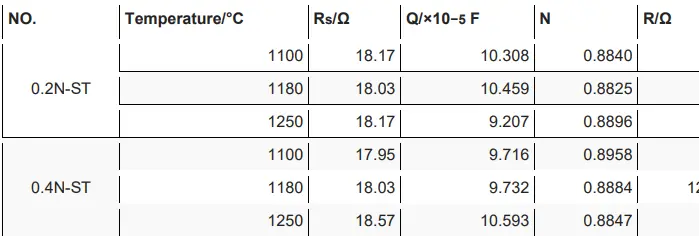
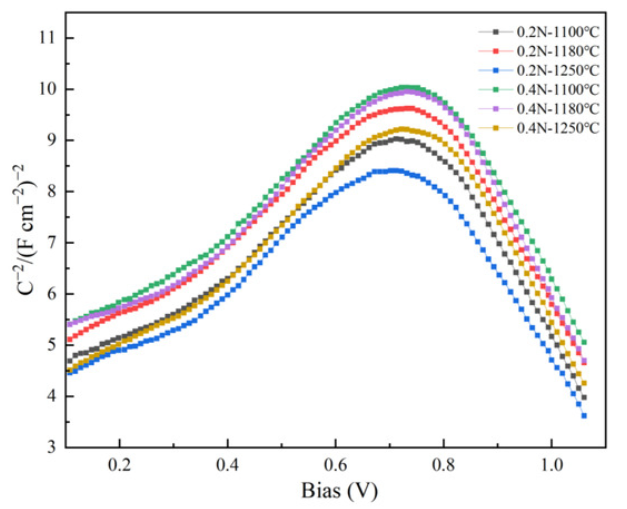
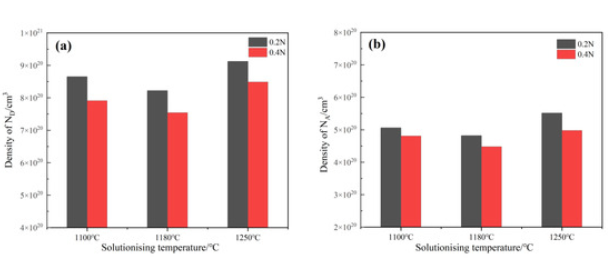
Mott–Schottky Analysis
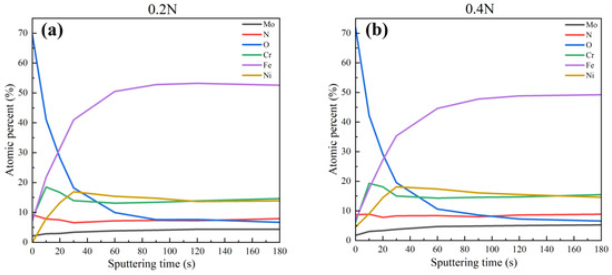
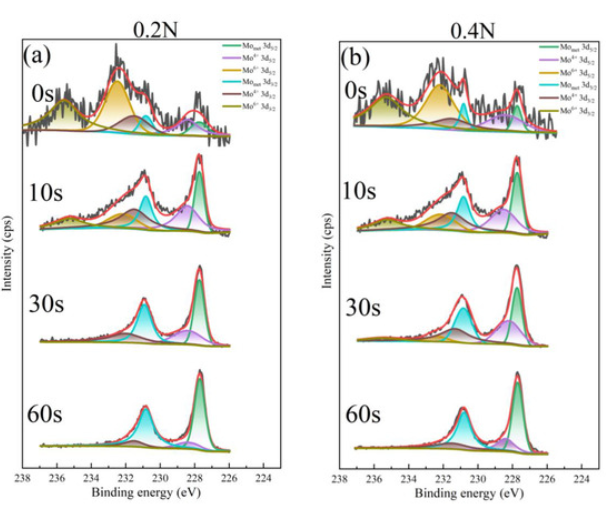
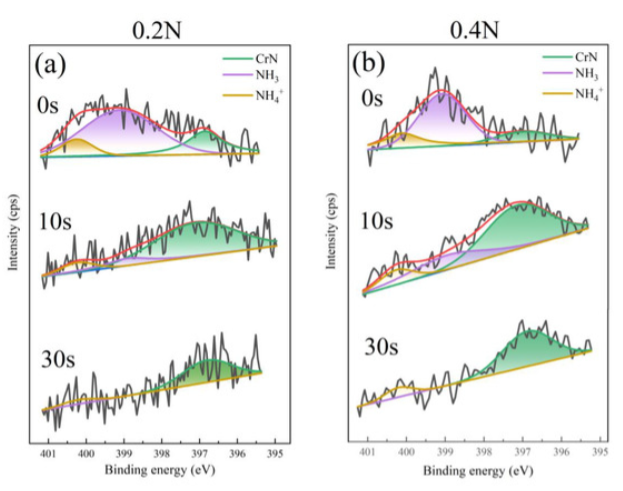
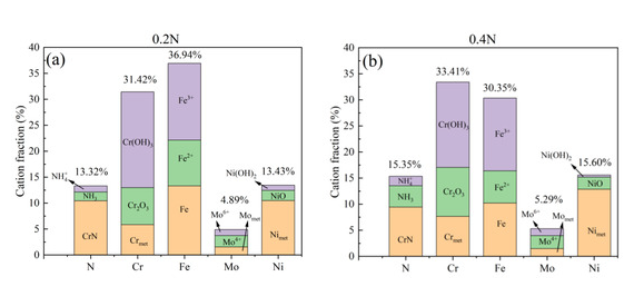
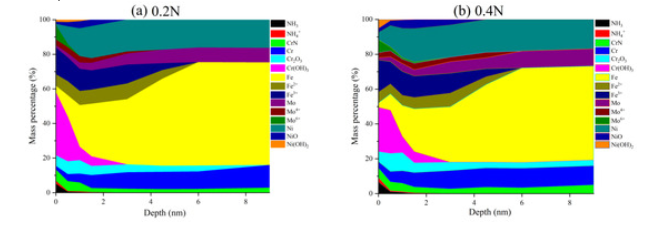
XPS Analysis