Request A Quote
Metal Corrosion and Hydrogen Permeation
Metals can absorb hydrogen (H) from corrosion processes, from welding, and from metal manufacture such as forming, coating, plating, and acid cleaning. The absorption and diffusion of atomic H in a metal can induce embrittlement which will seriously impair its corrosion resistance and mechanical properties. The harmful effects of hydrogen is influenced by its diffusivity rate and by the density of hydrogen traps in the host metal lattice. Irreversible H traps, such as oxides, require temperatures above 600 ºC for H to be released. These type of traps are sometimes used as means to capture H atoms to prevent their diffusion and suppress hydrogen-induced metal cracking.
Hydrogen permeation through metal alloys is measured with an electrochemical hydrogen permeation (HP) cell which has two compartments separated by a thin test specimen whose opposite surfaces are independently submerged in each of the two compartments. One compartment is the hydrogen charging side and the opposite compartment is the hydrogen exit side where the diffusing H atoms are detected, giving rise to a H permeation flux. Hydrogen permeation parameters are determined from permeation graphs to characterize embrittlement in metals. Hydrogen damage by metal cracking is subject to the type of alloy, the corroding environment, and the state of residual internal stress or externally applied stress to the alloy.
The propensity of H to enter and diffuse through a metal is measured by applying galvanostatic or potentiostatic electrolytic conditions to the metal in the charging compartment of the hydrogen permeation cell. Mild cathodic charging is necessary to generate a well defined hydrogen permeation transient curve to best characterize the hydrogen diffusion parameters. Free metal corrosion can induce hydrogen absorption, diffusion, and permeation through the metal from reduction reactions at its corroding surface. Also, galvanic corrosion can induce hydrogen absorption, diffusion, and permeation through the noble coupling metal.
The figure below shows the hydrogen permeation (HP) curve through a 500 µm thick iron (Fe) plate due to its galvanic coupling to zinc by electrically connecting both metals while simultaneously exposed to 0.1M H2SO4 solution in a HP cell. The galvanic coupling induced a cathodic galvanic current of 2.1 mA/cm2 generating hydrogen gas at the Fe metal surface, the more noble metal. A fraction of the generated hydrogen gas dissociated and absorbed as H atoms which diffused through the iron lattice until a steady H permeation flux was reached. A steady state H permeation flux through the iron plate is displayed as a steady state current close to 2µA/cm2, as displayed in the attached graph.
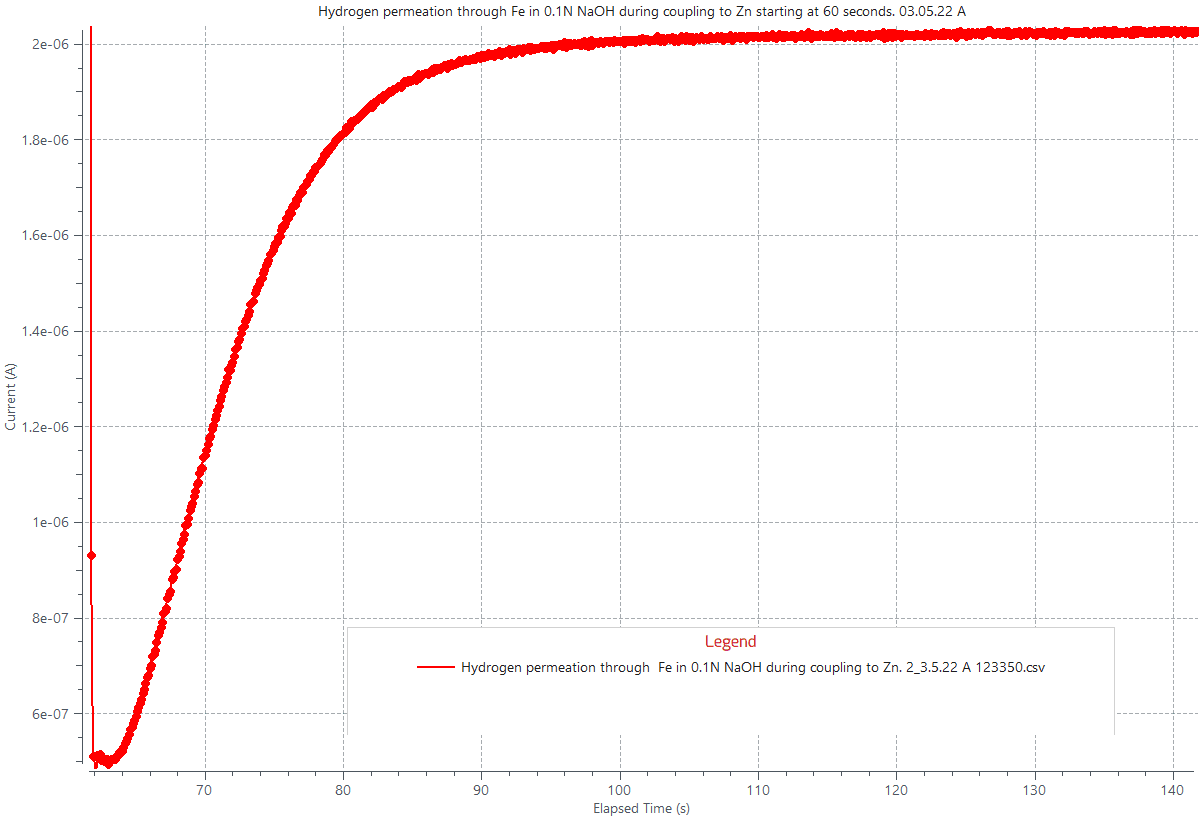
Hydrogen permeation through 1 cm2 area of iron plate, 500 µm thick, due to its galvanic coupling with zinc in 0.1M H2SO4 at room temperature. The coupling induced 2.1 mA/cm2 of cathodic current generating hydrogen gas at the iron surface, some of which absorbed as atomic H and diffused through the iron plate. A steady state H permeation flux through iron is displayed at the current plateau near the 2 µA/cm2 mark.

