

Services
Services on Metal Corrosion.
Aqueous or moisture corrosion involves an electrochemical and spontaneous electron transfer process. Such process is a result of simultaneous oxidation and reduction reactions at the corrosion site. At Corrosion Testing and Solutions, CT&S, we perform electrochemical corrosion testing as well as chemical corrosion testing by sample immersion in a corroding environment relevant to your industry. Testing is performed according to international standards. We also do customized corrosion testing to meet your unique process conditions. General and localized type of corrosion attack are evaluated.
The types of testing we do at CT&S include weight loss measurement, linear and Tafel polarization resistance, cyclic polarization, galvanostatic and potentiostatic polarization, corrosion inhibition, degree of sensitization due to carbide inter-granular precipitation, galvanic corrosion, electrochemical hydrogen permeation, and Electrochemical Impedance Spectroscopy or EIS. We determine corrosion parameters such as electrochemical corrosion potential, corrosion rate, corrosion current, ohmic resistance, polarization resistance, charge transfer resistance, passivation potential, pitting potential, re-passivation potential, critical pitting temperature, and galvanic corrosion risk failure.
We also have the capability to provide your team with electrochemical instrumentation and training on corrosion and testing methods. Having a testing capability to address corrosion issues on site is an economic and technical advantage. Corrosion equipments today are affordable, small in size, efficient, precise, and easy to perform on a table-top.
Electrochemical Impedance Spectroscopy (EIS)
Electrochemical Impedance Spectroscopy (EIS) is a non-destructive corrosion technique used to determine the metal impedance or resistance to current flow or electron transfer that takes place during electrochemical corrosion reactions. In this technique, a few mV of AC voltage is applied to the metal across a range of frequencies to generate an impedance spectrum. The impedance data is displayed as a Nyquist or Bode plot.
The metal’s ability to withstand corrosion attack arises from the impedance of its native oxide film separating the metal substrate from the corrosive electrolyte. The film impedance measures its resistance and reactance, that is, capacitance; both indicators of corrosion rate. A paint coating on the metal surface adds an additional resistance and capacitance component impacting corrosion rate. Coating capacitance is an indicator of coating integrity and barrier quality. Each of these surface impediments act to inhibit corrosion by suppressing electron transfer, the driving force of corrosion reactions.
The metal’s impedance data is used to extract corrosion parameters such as polarization resistance (Rp), needed to calculate corrosion rate; as well as charge transfer resistance (Rct), used also as an indicator of corrosion rate.
The side graph shows a Nyquist impedance spectrum from 304 stainless steel in a NaCl solution before and after anodic potentiodynamic cycling to potentials above the steel pitting potential. Prior to the potentiodynamic anodic cycling, the metal surface oxide is intact and provides protection against corrosion, resulting in an impedance spectrum as displayed by the dotted blue curve. After anodic polarization to potentials above the pitting potential, the oxide film breaks down giving rise to a suppressed impedance spectrums; as displayed by the red and blue solid lines contrasting with the dotted line generated prior to the potentiodynamic cycling.
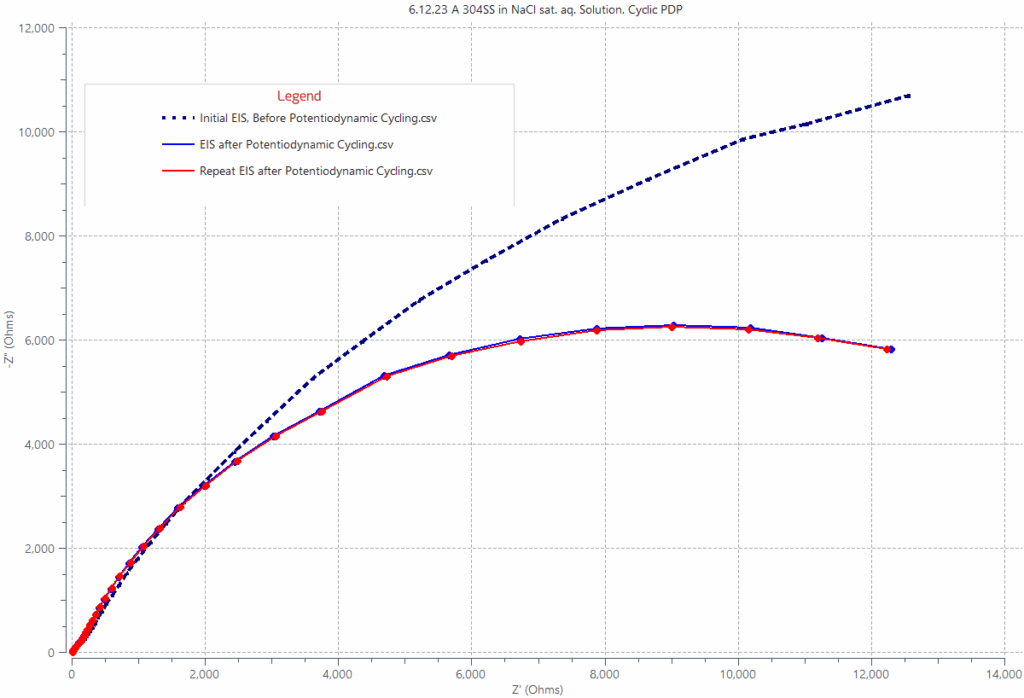
Nyquist plot or electrochemical impedance spectrum of 304 stainless steel in NaCl-saturated solution at open circuit (doted line) and after anodic polarization cycling beyond the steel pitting potential (red and blue solid lines). The impedance spectrums described by the dotted line and by the solid lines are clearly different, suggesting that the polarization cycling above the steel pitting potential changed the metal’s impedance significantly. The anodic polarization caused oxide breakdown of the native film which lead to a corresponding decrease in charge transfer resistance, realized from the depressed red/blue solid semi-circles relative to that of the dotted semi-circle obtained prior to the anodic polarization. The impedance data indicates that the breakdown of the passive film decreased the charge transfer resistance and made the steel less resistant to corrosion,
Galvanic Corrosion
Hydrogen Permeation
Hydrogen Embrittlement
Stress Corrosion Cracking
Inter Granular Corrosion
Pitting Corrosion
Crevice Corrosion
Uniform Corrosion
Disclaimer
The information contained in this website and on related work reports from Corrosion Testing & Solutions, LLC (CT&S) is for general information purposes only and does not constitute professional advice of any kind. The information is provided by Corrosion Testing & Solutions, LLC (CT&S). While we attempt to keep the information up to date and correct, we make no representations or warranties of any kind, express or implied, about the completeness, accuracy, reliability, suitability or availability with respect to the website or the information, products, services, or related graphics contained on the website or on related work reports from CT&S for any purpose. Any reliance you place on such information is at your own risk.

