Request A Quote
Hydrogen Embrittlement & Cracking in Metals: Steel, Copper & Stainless
Hydrogen Embrittlement Failure
Hydrogen embrittlement in metals is caused by the combined action of atomic hydrogen and residual or applied tensile stress to a metal. Embrittlement due to hydrogen (H) can lead to cracking and catastrophic brittle failures at stresses below the yield stress of susceptible materials. The most vulnerable are high-strength steels, titanium alloys, and aluminum alloys. Hydrogen embrittlement (HE) affects also alloys of nickel, copper, niobium, and vanadium. A typical example is stress corrosion cracking in steel pipelines. Fasteners used in bridges, aircraft, and ships are also known to be susceptible to HE. The effect is severe in the presence of hydrogen sulfide because H uptake by a metal increases in its presence.
Hydrogen can be introduced in metal parts from cathodic reactions that take place during free metal corrosion, during electroplating, during galvanic corrosion, and during cathodic protection practices. Hydrogen absorption can also occur from exposure to hydrogen-rich service environments such as hydrogen gas, hydrogen sulfide, and sour gas. Also, residual internal H from manufacture, welding, or pretreatment steps can lead to failure. Once absorbed, H diffuses quickly and collects at regions of high triaxial tensile stress where stress and H can build up and lead to metal embrittlement and sudden metal fracture.
Brittle fracture from embrittlement is characterized by clearly defined grain facets, as seen in the figure below, for an unstressed Ti-30Mo alloy plate subjected to electrolytic H cathodic charging at a high constant current of 15 mA/cm2 for 3.8 hours in 0.1N NaOH. The electrolytic introduction of H into metals at high rates can set up a high H concentration gradient able to induce lattice dislocations, dislocation movement, lattice deformation, and stress build up. The adjacent left bottom TEM micrograph displays dislocations piled up at grain boundaries after cathodic H charging at 3 mA/cm2 in 5% H2SO4. Plastic deformation induced by hydrogen loading into the metal lattice can also induce local deformation bands as a result of dislocation movement. In addition, the absorption, lattice diffusion, and recombination of hydrogen atoms will form hydrogen gas at defect lattice sites. Hydrogen gas pressure built up at these sites will form subsurface cavities or blisters at the metal surface. The attached right bottom micrograph exhibits such blisters surrounding a deformation band in a ti-30Mo alloy subjected to H cathodic charging at 25 mA/cm2.

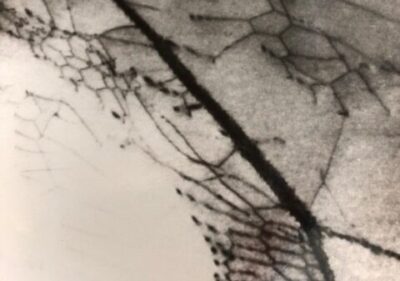
Ti -30Mo alloy showing network of dislocations piled up at a grain boundary caused by hydrogen diffusion from cathodic charging at 3 mA/cm2 in 5% H2SO4 for 2 hours. Magnification: 33,000X.

Ti -30Mo alloy plate exhibiting blisters when subjected to cathodic charging at 25 mA/cm2 in 5% H2SO4 for 20 hours. Magnification: 1300X.

