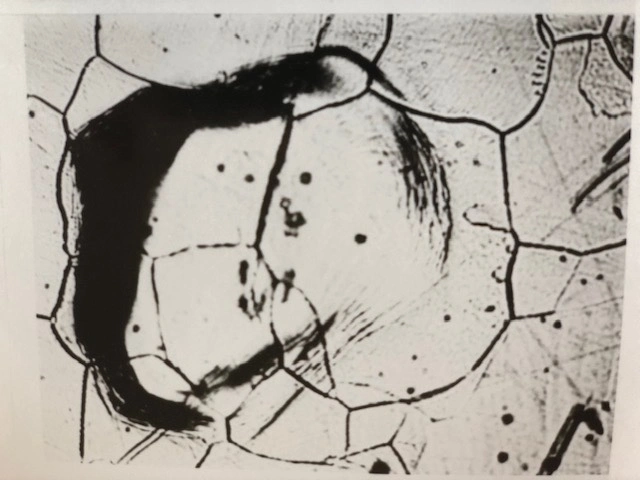
Back surface of iron plate, 500 µm thick, cathodically charged from its opposite front surface at 25 mA/cm2 in 5% H2SO4 for 5 hours at room temperature. Hydrogen loading by the cathodic charging caused the observed lattice deformation and slip lines at the back iron surface. No external stress was applied. 200X.
Corrosion Testing and Characterization
Surface Characterization
The corrosion of a metal alloy is impacted by its metallurgical state, its surface condition, its electrochemical corrosion potential in the corroding medium, as well as the corroding environment conditions such as composition, temperature, pH, and the content of oxidizing agents including oxygen, chloride, and sulfide. The surface degradation and form of attack and failure from corrosion reactions can be characterized by optical or scanning electron microscopy and by Energy Dispersive X-Ray Spectroscopy, which is used to determine the surface composition of the corroded surface.
Typical degradations by corrosion are surface oxidation, metal thinning, and pitting. Also, corrosion reactions can lead to some loss of material strength due to hydrogen absorption leading to metal embrittlement. The sole absorption of hydrogen can induce metal deformation in the absence of applied stress. Metals absorb hydrogen from free corrosion, galvanic corrosion, electrolytic surface pre-treatments, and from cathodic protection practices done to control corrosion.
Heavy introduction of hydrogen into a metal can induce high hydrogen concentration gradients as well as dislocation generation and dislocation movement, plastically deforming the metal lattice. The adjacent optical micrograph shows the back surface of an iron plate cathodically charged with hydrogen from its opposite surface at 25 mA/cm2 in 5% sulfuric acid. The charging caused lattice deformation and slip lines at the metal surface opposite to the charging side. Slip lines result from the sliding of block of crystals along slip planes induced by the deformation.
Corrosion Measurement
Metal alloys corrode at different rates subject to the type of alloy and the corroding environment. Localized type of corrosion conducing to pitting occurs at a much higher rate than uniform corrosion which takes place over the entire metal surface. Pitting on metals such as stainless steels, aluminum, copper, or nickel can be characterized by the magnitude of their respective pitting potentials in a given corroding medium. Uniform corrosion rate can be determined by immersing a metal sample in the respective corroding medium and measuring the metal weight loss over time.
Uniform corrosion can also be measured electrochemically by polarizating the metal sample through cathodic and anodic potentials relative to the corrosion potential of the metal, Ecorr. The polarization generates a linear cathodic and a linear anodic region below and above Ecorr, respectively, whose intersection leads to determining the corrosion current density, Icorr. The corrosion rate is proportional to Icorr. It is calculated from Icorr and from parameters that are intrinsic to the corroding metal alloy.
The side graphic data is an example for copper and 303 stainless steel, 303ss, in 3.5% NaCl. The graphical data reveals an Icorr for copper (Icorr Cu) which is larger than that of 303SS (Icorr 303SS) by more than one order of magnitude. The much lower corrosion rate of the stainless steel sample is due to its native protective surface oxide film, unlike the case of copper which forms a less protective and more porous oxide film on its surface.


