Electrochemical Corrosion Testing Services
Metal corrosion can be evaluated electrochemically or by a chemical immersion method. The electrochemical method is a fast and reliable technique. This method determines the electrochemical behavior of a metal sample in a solution of known temperature, pH, and composition. Corrosion by chemical immersion in a known solution is done by measuring the sample weight loss over time in hours, days, or weeks; guided by the ASTM G31 standard.
We evaluate the corrosion characteristics of ferrous and non-ferrous metal alloys determined by the anodic and the cathodic reactions taking place at the corroding surface. The anodic reaction causes metal dissolution and weight loss in alloys that form a porous oxide film, or localized pitting attack in alloys protected by a compact oxide film which acts as a barrier against the corroding environment. In some corroding environments, the cathodic reactions can lead to hydrogen uptake by the metal, detrimental under residual or applied stress when exposed to chloride, carbon dioxide, or hydrogen sulfide.
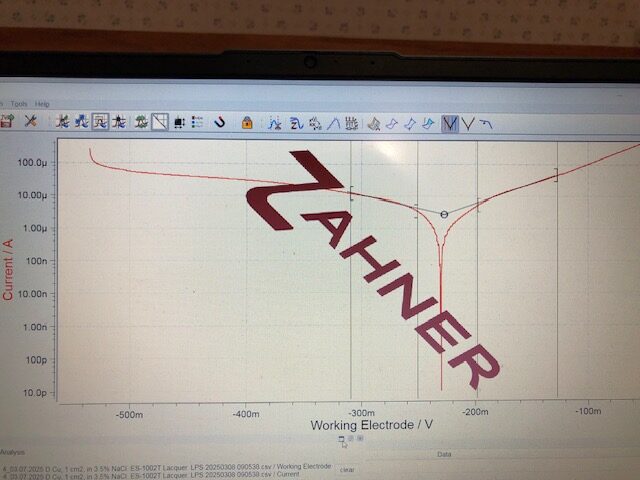
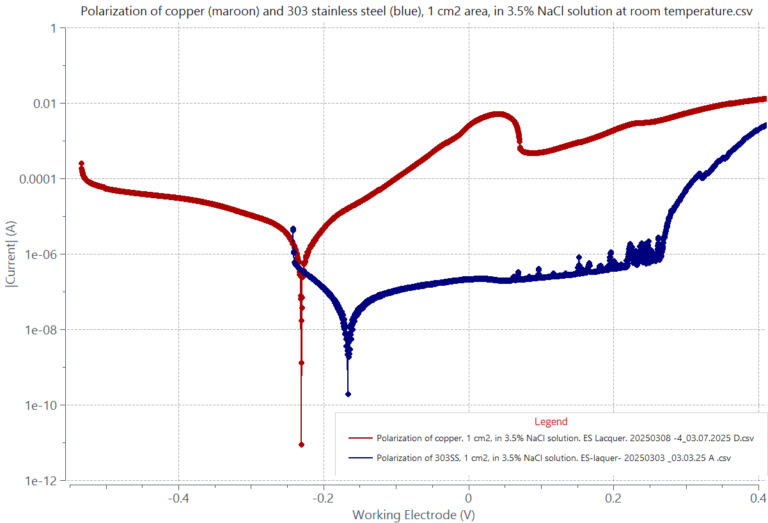
The resistance to corrosion is subject to the type of metal alloy and its corroding environment. Copper and copper alloys do not form a protective oxide film and corrode easily by metal dissolution when exposed to an aggressive marine ambient. The left graph shows the electrochemical polarization behavior of copper in a marine-like solution containing 3.5% NaCl, sodium chloride. Analysis of the linear graph below and above the copper corrosion potential of -0.228 V provides a corrosion dissolution rate of 26.7 µm/y.
Examples of alloys that resist to corrosion are stainless steels. The adjacent polarization graph displays a blue curve corresponding to the polarization of 303 stainless steel (SS) and a red curve corresponding to the polarization of copper in 3.5% NaCl solution at room temperature. The anodic dissolution region of 303 SS from -0.125 V to about +0.265 V exhibits a significantly suppressed metal dissolution current, unlike the case of copper. Accordingly, the corrosion rate of 303 SS is suppressed to about 0.626 µA/y. The observed small current spikes starting at 0.061 V are due to a breakdown/repassivation event of the SS surface caused by the action of chloride. Oxide breakdown and re-passivation events take place until the steel pitting potential is reached at about 0.265 V. Beyond the pitting potential the alloy is unable to re-passivate, causing a fast and continuous current rise due to pitting corrosion.

Polarization linear sweep of copper, 1 cm2, in 3.5% NaCl solution at room temperature displaying a corrosion potential at -0.228 V vs. Ag/AgCl. The steep slope above -0.228 V indicates large anodic currents due to its fast corrosion rate calculated at 26.7 µm/y.

Red-Cu: Uninhibited Corrosion. No Pitting Blue-303SS: Inhibited Corrosion. Pitting Polarization of 303 SS, 1 cm2, in 3.5% NaCl solution (blue) showing a suppressed current of about 0.626 µA/y between -0.166V and 0.265V, contrasting that of copper (red). Starting at 0.265 V, 303SS, exhibits a large current increase due to a breakdown of the oxide film leading to pitting attack.

Pits formed on 304 stainless steel after polarizing this alloy in 3.5% NaCl solution to potentials beyond its pitting potential.

