

Corrosion
Importance of Corrosion Testing.
Industries that use metallic materials in mild or aggressive environments rely on the metal’s corrosion performance. Generally, materials specifications on metal alloys for use in most industries include chemical and metallurgical composition as well as physical and mechanical properties. The corresponding corrosion resistance requirements are sometimes lacking or incomplete. Testing a material’s behavior in a given working corroding environment will determine its suitability for such environment.
Testing the conditions to which your alloys are to be exposed will lead to selecting a proper alloy. Factors impacting metal corrosion are type of alloy, treatment history, metallurgical composition, pH, composition of the corroding environment, temperature, and state of internal residual stress or externally applied stress to the metal.
The attached graphical electrochemical corrosion data was generated using a hydrogen permeation electrochemical cell holding an iron (Fe) sample separating two compartments, one anodic and the other cathodic. The graph shows current decays, due to oxidation reactions taking place on the iron surface facing the anodic compartment containing 0.1N NaOH. Current rises in the graph were caused by reduction reactions on the iron surface facing the cathodic compartment containing 0.1M H2SO4. Hydrogen (H) is generated in the cathodic compartment by galvanic corrosion or by free corrosion of the iron surface. In the opposite iron surface in contact with 0.1N NaOH, an oxidizing anodic potential is applied to measure the H flux permeating through the iron plate due to its galvanic coupling to zinc (Zn) or due to the iron free corrosion. The magnitude of the current rise to a steady state value indicates the rate at which hydrogen is diffusing through the metal membrane.
Assessing the metal’s corrosion response to its environment is essential and beneficial to many industries relying on metals including manufacturing, chemical, electrochemical, petrochemical, oil, refinery, construction, transportation, automotive, aerospace, energy, water cooling systems, utility, mining, electronics, food, health, and medical industry. At Corrosion Testing & Solutions, LLC (CT&S), we will test your materials in environments relevant to your production process conditions.
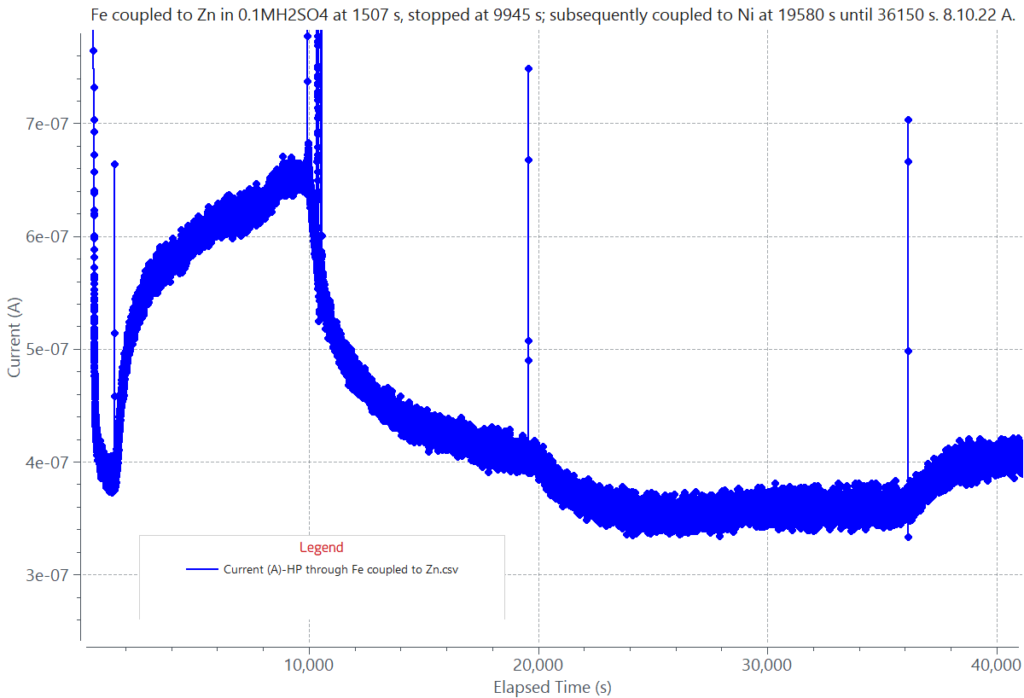

Oxidation current decays and reduction current rises data from iron (Fe) due to: a) Fe coupling to Zn, causing H generation and its permeation through Fe (1507 s to 9945 s); b) Fe coupling to Ni, causing oxidation of Fe and a current decay (19580 s to 36150 s); and c) Fe free corrosion in 0.1M H2SO4 (36150 s to 42000 s) after stopping Fe coupling to Ni at 36150 s. The initial fast current decay to about 400 nA is due to an oxidation potential applied to Fe exposed to 0.1N NaOH done to measure the H flux through Fe during stages (a) or (c). The current decay from 9945 s to 19580 s is due to the decrease in H flux through iron, after stopping the Fe-Zn coupling at 9945 s.

