Corrosion Issues and Testing Solutions
All metal alloys are prone to be degraded when they are exposed to a corroding medium or to adverse environmental factors including moisture, oxygen, and salt. At Corrosion Testing & Solutions, LLC (CT&S), we do corrosion testing and provide training to help your business address corrosion issues. Protection against corrosion failure starts with testing and selecting suitable metal alloys for best service in a given corroding environment. Our services are oriented to all industries that process or use metal alloys exposed to corroding aqueous or moist environments.
Corrosion is caused by oxidation and reduction reactions taking place at the corroding surface in contact with the corroding medium. These two types of reactions can independently lead to failure. The oxidation reactions cause metal thinning or localized attack, subject to the type of alloy and the corroding conditions. The reduction reactions can cause harmful hydrogen absorption by the corroding metal in acidic or alkaline environments. The absorption and diffusion of hydrogen into the corroding metal can induce lattice defects, lattice deformation, and stress build up; which can result in metal embrittlement and sudden failure in some alloys.
At CT&S we are equipped to evaluate the corrosion performance of ferrous and nonferrous alloys simulating your corroding medium using electrochemical techniques to determine corrosion rate and type of corrosion attack. This technique is fast and very reliable. Longer term immersion corrosion testing using ASTM G31 is also performed.
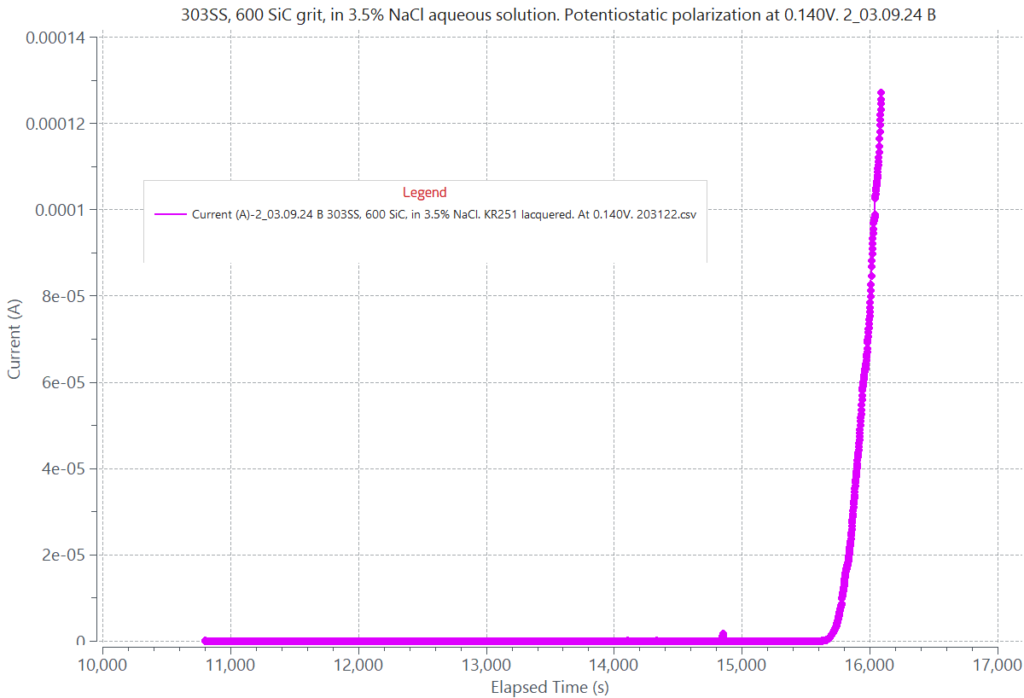

Graph displaying pitting corrosion of 303 stainless steel (SS) in 3.5% NaCl aqueous solution during pototentiostatic polarization at 0.140V above its corrosion potential. A sudden sharp and continuous current rise is seen at 4900 seconds from the start of polarization. The rise in current is due to a breakdown of the protective oxide film on 303 SS caused by chloride. leading to pitting corrosion attack.

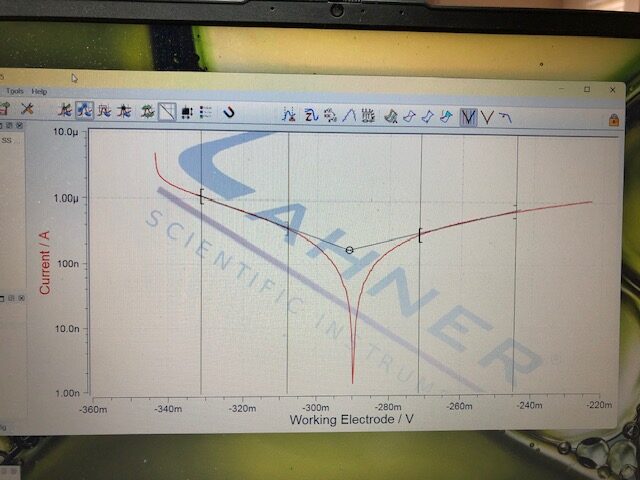
Graph generated from corrosion of 303 stailess steel in NaCl-saturated aqueous solution. The corresponding uniform corrosion rate determined by Tafel polarization is 1.44 µA/cm2 at room temperature. The chloride content in the corroding solution will induce pitting corrosion causing a metal dissolution rate much higher than 1.44 µA/cm2.

