Request A Quote
Crevice Corrosion of passive metals: Alloys of Aluminum, Nickel, Titanium, and Stainless Steels
Crevice Corrosion Failure
Crevice corrosion is a localized attack that occurs in occluded areas at metal/metal or metal/nonmetal tight junctions exposed to a corroding stagnant electrolyte with limited air circulation. This type of attack is found in unsealed joints, in threaded connections, under surface deposits, and under gaskets, washers, teflon, clamps, flanges, bolts, or nuts. Crevice corrosion is prevalent in metals and alloys that develop a native protective oxide film. The crevice environment at the occluded site can reach high acidification causing local breakdown of the barrier protective oxide film prompting pitting attack at this site. Crevice corrosion at bolts and flange joints of stainless steel is common in marine environments. This type of corrosion can be suppressed by good material selection, minimizing creviced designs having bolted joints, or applying a proper coating at metal-metal or metal-nonmetal tight junctions.
The graph Current vs. Working Electrode Voltage shown below was generated according to standard ASTM G61. It displays crevice corrosion data collected during potentiodynamic anodic polarization of 304 stainless steel (SS) in pure distilled water. The crevice corrosion took place at the interface joining the 304 SS round sample and the epoxy resin mount holding the sample. The sudden and continuous increase in anodic current starting at about 0.680 V was due to crevice corrosion.

Typical examples of Crevice Corrosion attack in 316 stainless steel steam condenser at tight metal junctions accessed by warm seawater. The attack was enhanced by the presence of chloride ions in the seawater whose composition is expected to contain about 3.5% NaCl (2.12% of chloride).
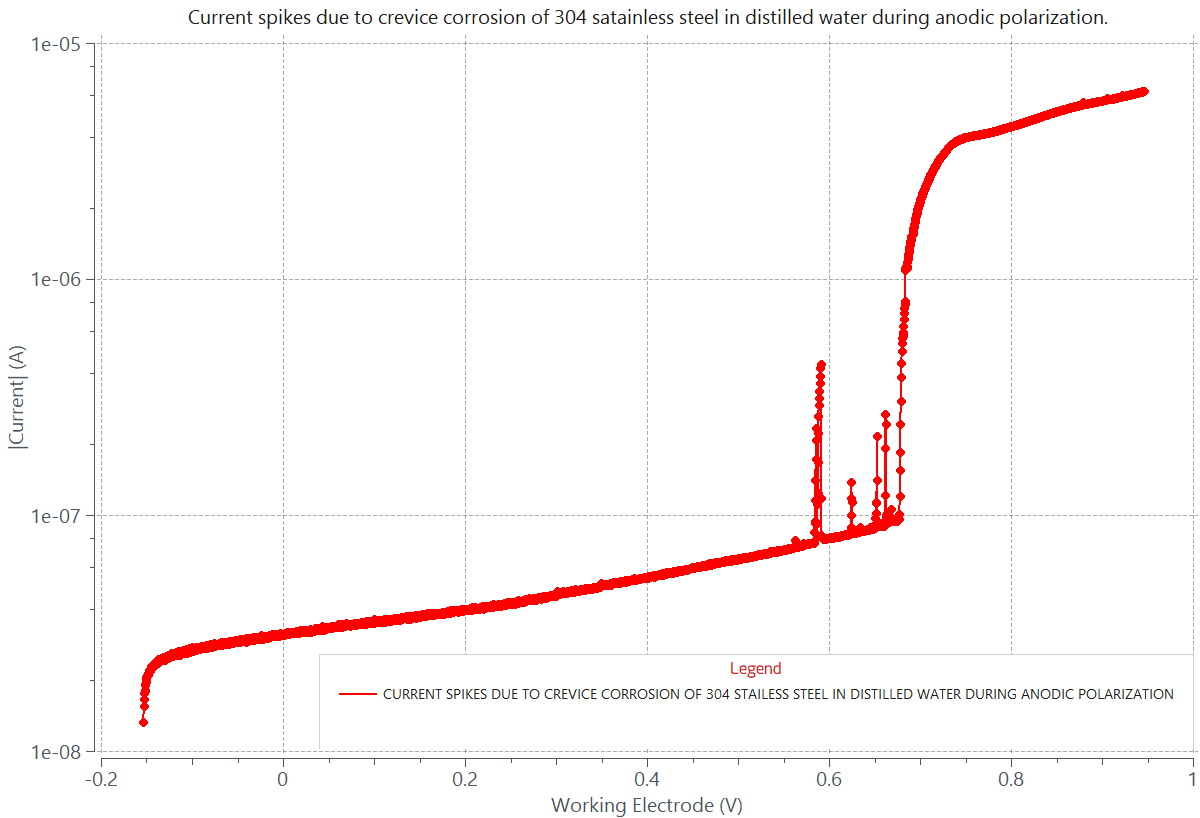
Crevice corrosion attack of 304 stainless steel (1 cm2) in pure distilled water at room temperature. The graph displays a sudden and continuous increase in anodic current at about 0.680V, relative to a Ag/AgCl reference electrode. The continuous rise in current is due to crevice corrosion attack at the area interface between the round metal sample and the surrounding epoxy resin holding the metal sample.

