HYDROGEN EMBRITTLEMENT
Hydrogen embrittlement (HE) is caused by the combined action of hydrogen and residual or applied tensile stress to a metal. Embrittlement due to hydrogen (H) can lead to cracking and catastrophic brittle failures at stresses below the yield stress of susceptible materials. The most vulnerable are high-strength steels, titanium alloys, and aluminum alloys. HE affects also alloys of nickel, copper, niobium, and vanadium. A typical example is stress corrosion cracking in steel pipelines. The effect is most severe in the presence of hydrogen sulfide because H uptake by the metal increases in the presence of hydrogen sulfide. Cracking failures due to HE is also well known in high strength threaded steel fasteners used in bridges, vehicle engines, aircraft, and ships.
Hydrogen can be introduced in metal parts from cathodic reactions that take place during free metal corrosion, during electroplating, during galvanic corrosion, and during cathodic protection practices. Also, residual internal H from manufacture, welding, or pretreatment steps can lead to failure. Once absorbed, H diffuses quickly and collects at regions of high triaxial tensile stress where stress and H can built up leading to metal embrittlement and sudden metal fracture.
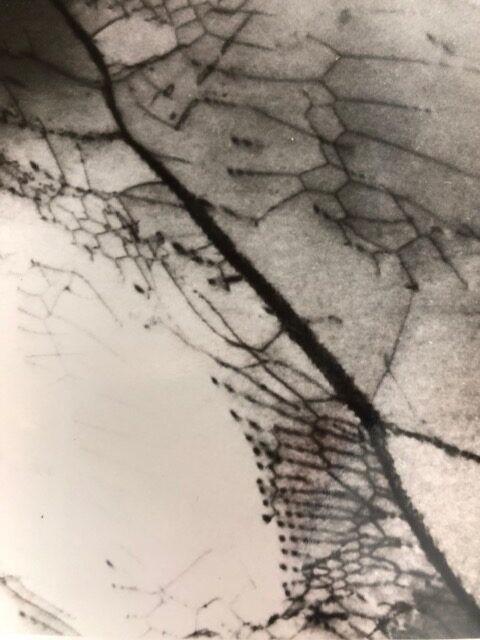
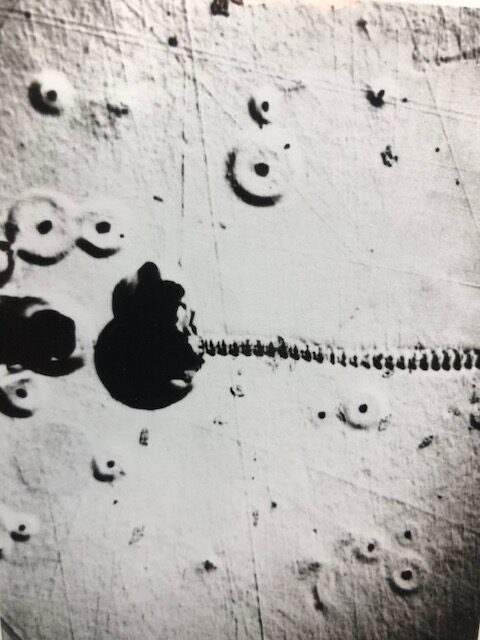
Brittle fracture from embrittlement is characterized by clearly defined grain facets, as seen in the figure below, for an unstressed Ti-30Mo alloy plate subjected to H cathodic charging at a high constant current of 15 mA/cm2 for 3.8 hours in 0.1N NaOH. The introduction of H into metals at high rates can set up a high H concentration gradient able to induce lattice dislocations and lattice deformation aided by dislocation movement. The adjacent figures below display dislocations piled up at grain boundaries and surface blisters generated from hydrogen cathodic loading into Ti-30Mo alloy, in the absence of applied stress. Blisters are subsurface cavities formed by H lattice diffusion and recombination at defect sites generating hydrogen gas which exerts pressure build up within the cavity.
The procedure to evaluate hydrogen uptake, permeation, and transport in metals is done according to the standard practice ASTM G148.

Brittle fracture along the thickness of a 550 µm thick Ti alloy plate in the absence of applied stress caused only by hydrogen cathodic charging at 15 mA/cm2 for 3.8 hours in 0.1N NaOH. Clearly defined grain facets are observed throughout the thickness of the fractured surface.